Mexico has cleared Qdenga, Takeda’s dengue vaccine, but the approval raises almost as many questions as it answers. The decision gives the country a new regulatory tool against a mosquito-borne disease that has surged hard in recent years. It does not, by itself, mean a national vaccination campaign starts tomorrow. What matters next is who will get the vaccine first, how it will be distributed, and whether health authorities use it in the areas carrying the heaviest dengue burden.
What the approval changes
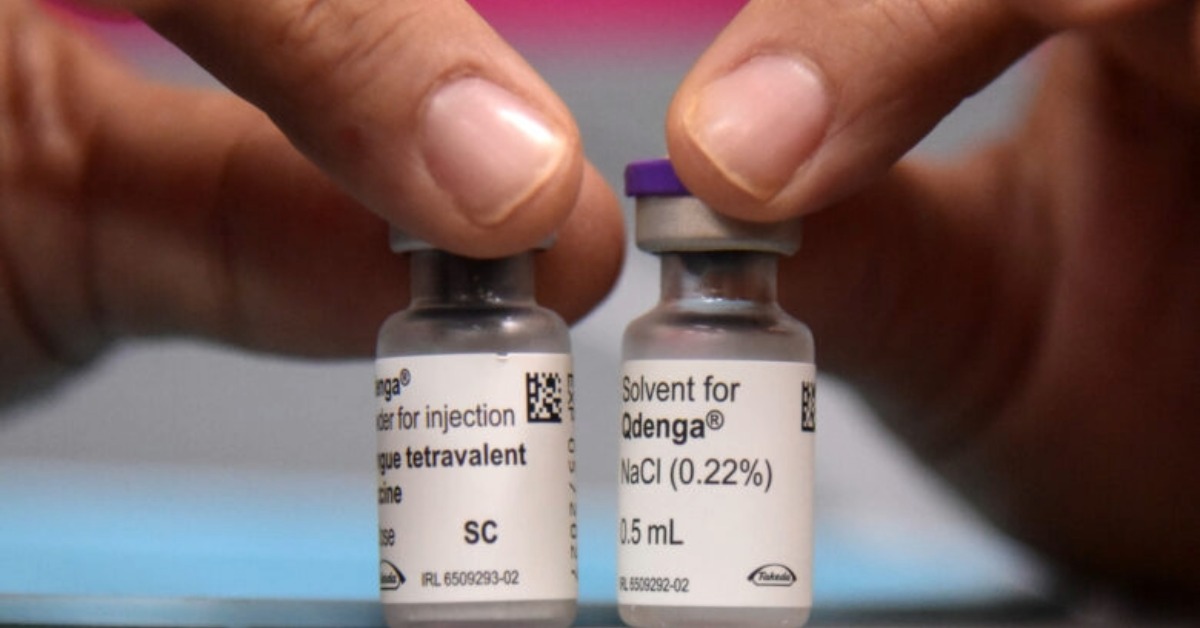
Mexico’s health regulator, Cofepris, has cleared Qdenga, the dengue vaccine developed by Takeda, for use in Mexico. That matters because dengue is no longer a limited seasonal problem in many states. It has become a serious public health threat. Patients may need hospitalization, and severe cases can turn deadly. The decision gives doctors and health planners another prevention tool. That is important at a time when mosquito control has not stopped repeated outbreaks. The move is also a regulatory step, not a full rollout. Approval can open the market, but it does not create a public campaign on its own. It also does not guarantee immediate access in clinics or pharmacies. That distinction will shape what families see next. It will also shape how quickly providers respond. For residents in Mexico, especially in warm and humid areas, the message is clear. Prevention options may widen beyond repellents, screens, fumigation, and cleanup drives.
What Qdenga is and how it is used
Qdenga is a live-attenuated vaccine built to protect against the four known dengue serotypes. That broad scope matters. A first infection does not prevent later infection with a different serotype. In some cases, repeat infections can raise the risk of severe disease. International guidance treats the vaccine as a targeted public health tool. It is not a universal answer for everyone. The World Health Organization recommends its use in children ages 6 to 16 in areas with high transmission. The schedule is two doses given three months apart. WHO also says vaccination should sit beside vector control and public education. It is not meant to replace them. That point matters in Mexico. Regulatory clearance opens the door, but authorities still must decide who gets priority. They must also decide whether the vaccine will enter public programs, high-burden regions, or the private market first. Doctors will also have to explain who is likely to benefit most.
Why the timing matters
The timing stands out because Mexico has faced a heavy dengue burden in recent years. Reports reviewed for this article say the country recorded more than 125,000 cases and 478 deaths in 2024. In 2025, the total fell sharply, but Mexico still logged nearly 22,000 cases and 85 deaths. Early 2026 data showed 896 confirmed cases and no confirmed deaths when published. Official surveillance also found that most early 2026 cases were concentrated in Sonora, Sinaloa, Veracruz, Tabasco, and Baja California Sur. That concentration helps explain why vaccine policy matters beyond the health sector. Dengue can affect schools, workplaces, local tourism, and household budgets. It also puts pressure on clinics and hospitals during periods of higher transmission. For foreigners who live in Mexico year-round or for long stretches, the risk is not theoretical. In many communities, it is part of daily life during mosquito season and sometimes well beyond it.
The questions that come next
Takeda says Qdenga has been authorized in 41 countries. The company also said 18.6 million doses had been distributed in 11 endemic countries as of September 2025. That wider record gives Mexico more evidence than earlier adopters had. Still, the next story is not the approval itself. It is access. Families will want to know when doses will arrive, where they will be offered, and how much they will cost. They will also want to know who will be eligible first. Those answers will shape whether Qdenga remains largely private or adopts a broader public strategy in higher-risk areas. For readers living in Mexico, the most practical takeaway is simple. The approval matters, but it does not replace basic protection. Mosquito control, standing-water cleanup, and window screens still matter. So does fast medical attention for warning signs while authorities and providers decide how this vaccine will actually reach patients.
With information from Takeda, Secretaría de Salud